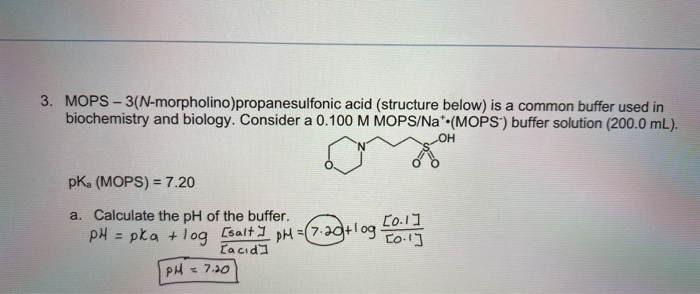
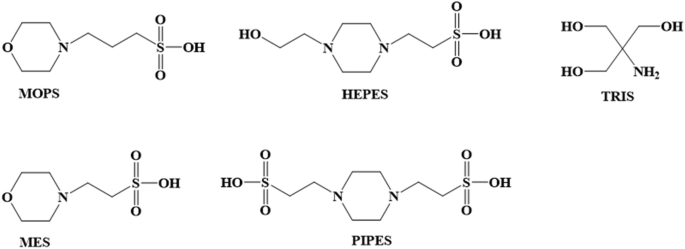
You prepare a buffer solution from 10.0 mL of 0.100 M MOPS (3- morpholinopropane-1-sulfonic acid) and 10.0 mL of 0.077 M NaOH. Next, you add 1.00 mL of 2.05 x 10^-5 M
Not just a background: pH buffers do interact with lanthanide ions—a Europium(III) case study | SpringerLink
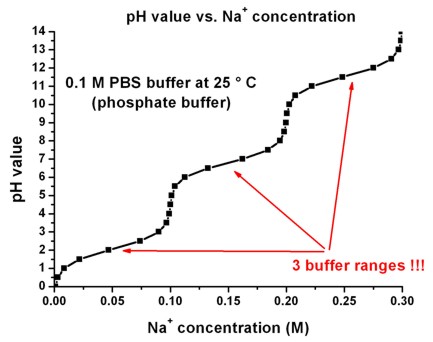
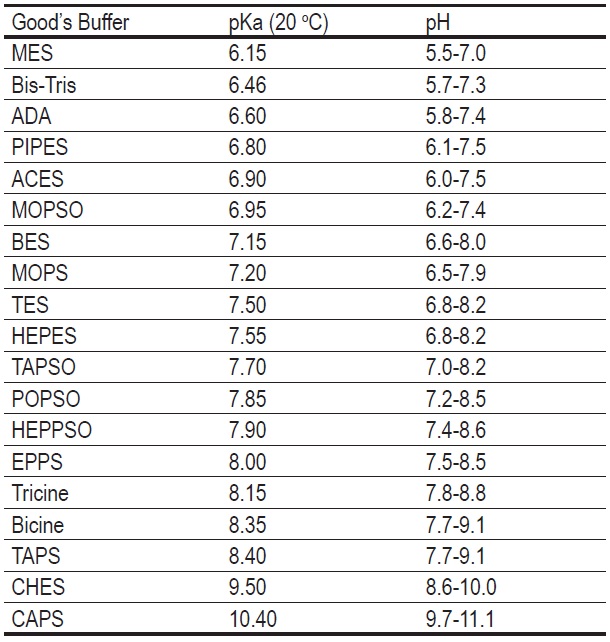
Un)suitability of the use of pH buffers in biological, biochemical and environmental studies and their interaction with metal i
Un)suitability of the use of pH buffers in biological, biochemical and environmental studies and their interaction with metal i

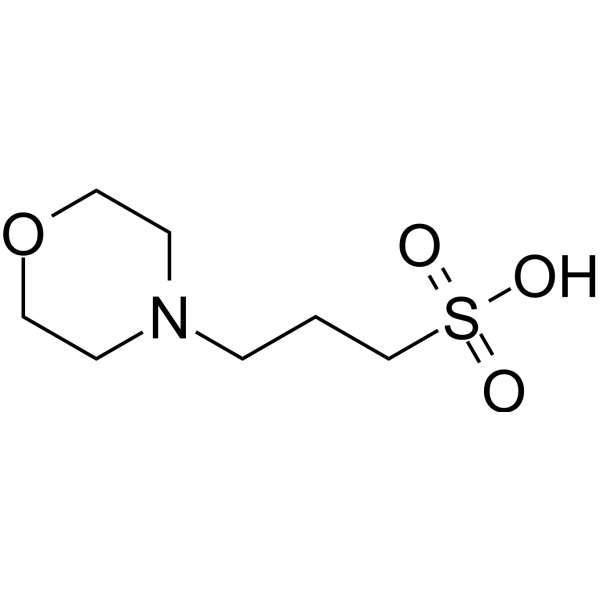
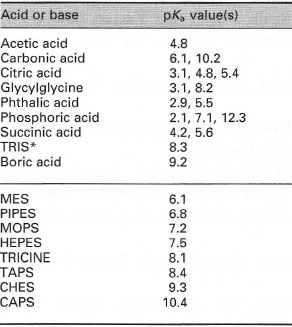
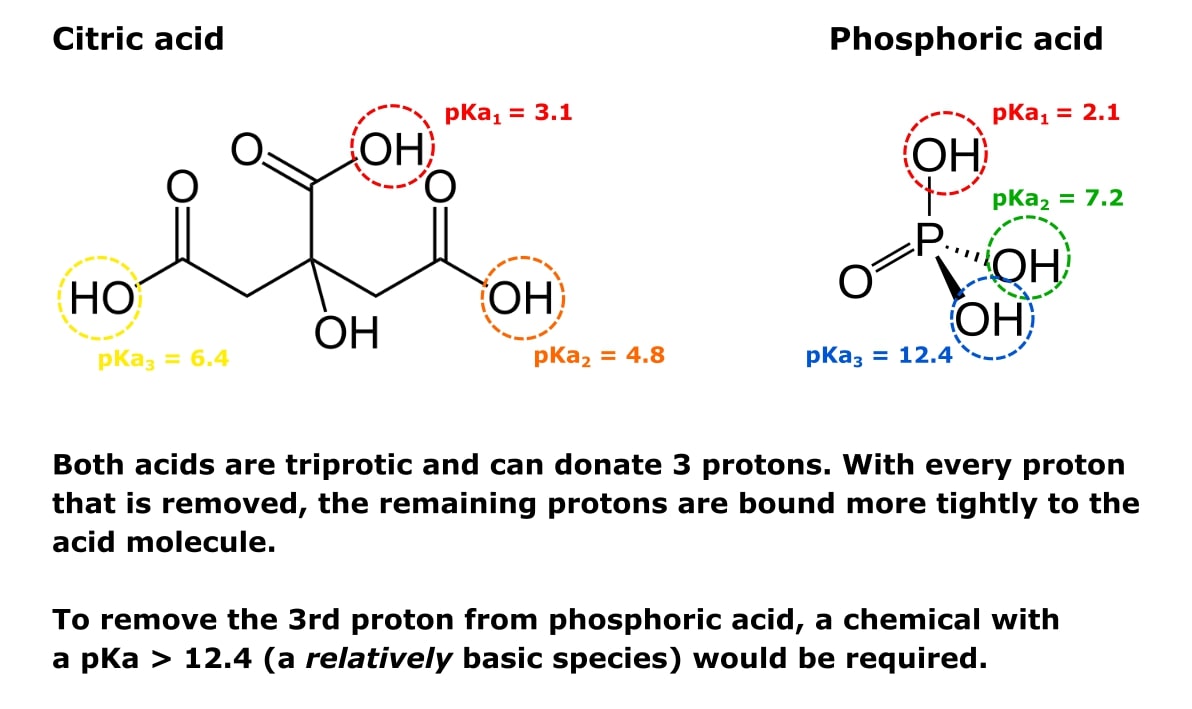

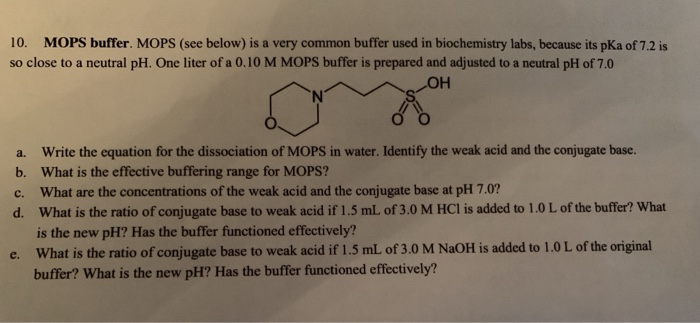

![MOPS Buffer [3-(N-Morpholinopropanesulfonic Acid] - 1132-61-2 - Discovery Fine Chemicals MOPS Buffer [3-(N-Morpholinopropanesulfonic Acid] - 1132-61-2 - Discovery Fine Chemicals](https://discofinechem.com/wp-content/uploads/2015/08/1132-61-2.png)