Newaner 500 ml stainless steel double mesh milk frother manual, 8 x 8 x 18 cm. Perfect milk foam thanks to double sieve and special press mechanism : Amazon.co.uk: Home & Kitchen

How evolution shapes enzyme selectivity – lessons from aminoacyl‐tRNA synthetases and other amino acid utilizing enzymes - Tawfik - 2020 - The FEBS Journal - Wiley Online Library
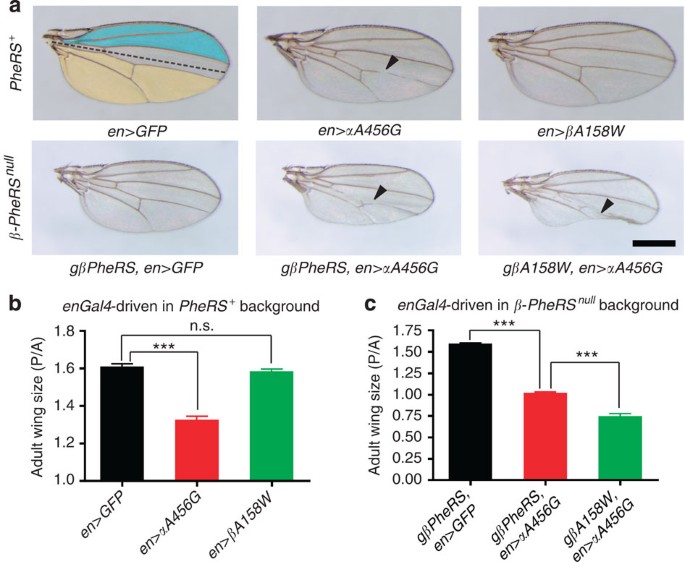
Double-sieving-defective aminoacyl-tRNA synthetase causes protein mistranslation and affects cellular physiology and development | Nature Communications
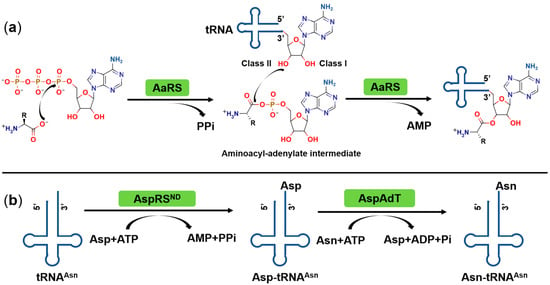
IJMS | Free Full-Text | Aminoacyl-tRNA Synthetases as Valuable Targets for Antimicrobial Drug Discovery

Fluoride removal from water by using micron zirconia/zeolite molecular sieve: Characterization and mechanism - ScienceDirect

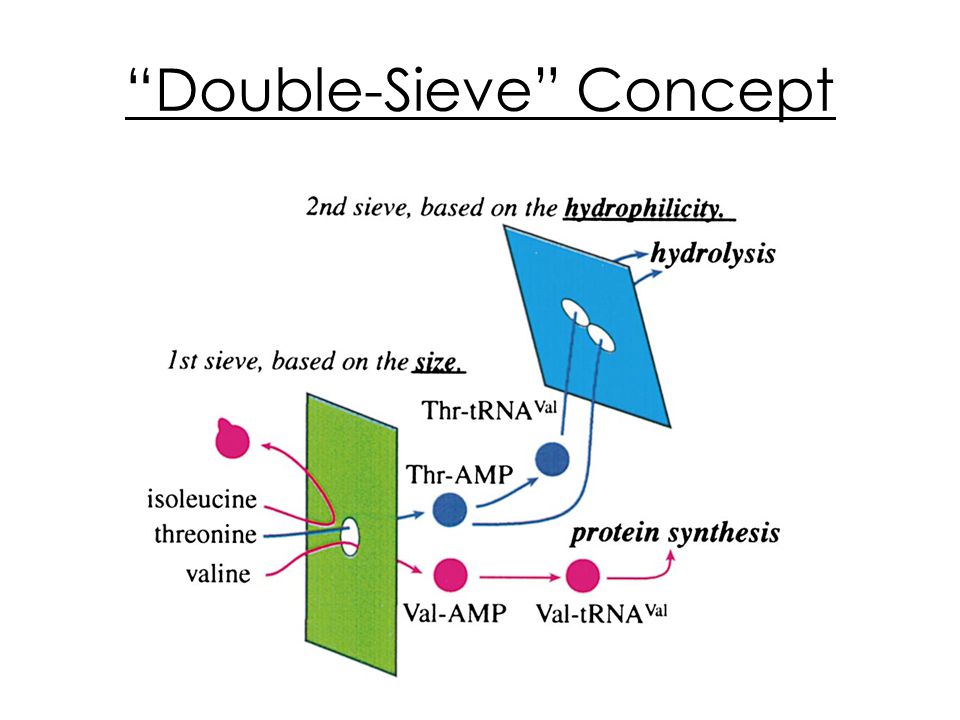
Structural Basis for Double-Sieve Discrimination of L-Valine from L-Isoleucine and L-Threonine by the Complex of tRNAVal and Valyl-tRNA Synthetase: Cell

Severe oxidative stress induces protein mistranslation through impairment of an aminoacyl-tRNA synthetase editing site | PNAS
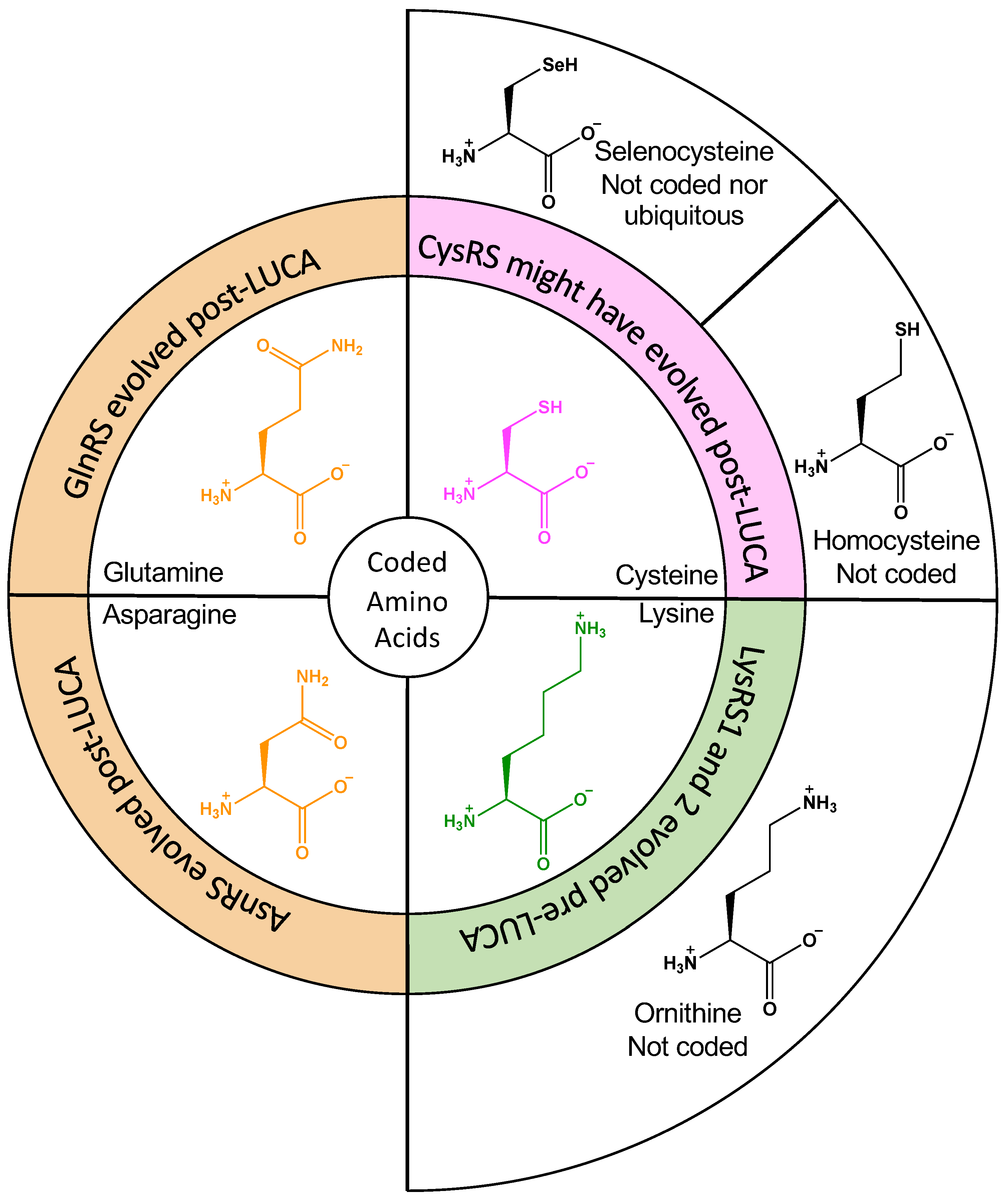
Genes | Free Full-Text | Did Amino Acid Side Chain Reactivity Dictate the Composition and Timing of Aminoacyl-tRNA Synthetase Evolution?

The binding mode of orphan glycyl-tRNA synthetase with tRNA supports the synthetase classification and reveals large domain movements | Science Advances

Optimal multi-depot location decision using particle swarm optimization - Yin-Mou Shen, Ruey-Maw Chen, 2017

Double-sieving-defective aminoacyl-tRNA synthetase causes protein mistranslation and affects cellular physiology and development | Nature Communications

Figure 5.7 from Revisiting the 'Double-Sieve Model' for editing mechanism in aaRSs | Semantic Scholar

Structural Basis for Double-Sieve Discrimination of L-Valine from L-Isoleucine and L-Threonine by the Complex of tRNAVal and Valyl-tRNA Synthetase: Cell

Structural Basis for Double-Sieve Discrimination of L-Valine from L-Isoleucine and L-Threonine by the Complex of tRNAVal and Valyl-tRNA Synthetase: Cell
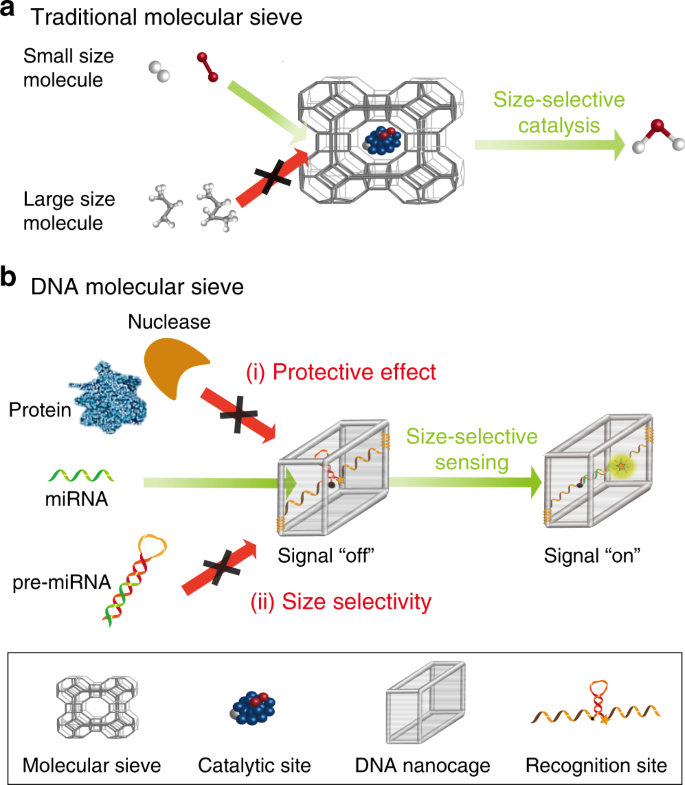
Size-selective molecular recognition based on a confined DNA molecular sieve using cavity-tunable framework nucleic acids | Nature Communications

SOLVED: lleRS users a double-sieve mechanism to accurately produce lle-tRNA^11e and prevent the synthesis of Val-tRNA^11e. Which order pairs of amino acids differ in structure by a single carbon and might have

Figure 5.5 from Revisiting the 'Double-Sieve Model' for editing mechanism in aaRSs | Semantic Scholar

Nanoconfining Cation-π Interactions as a Modular Strategy to Construct Injectable Self-Healing Hydrogel | CCS Chemistry