a) Cobalt magnetic moment in the cubic CAFM magnetic structure as a... | Download Scientific Diagram

SOLVED: Problem 6. A graduate student managed to prepare a red-brown complex compound of cobalt with the Schiff-base ligand shown below. Calculate the magnetic moment of the Co(salen) complex (CHCoNO) (Mol. Wt.:
transition metals - How to compare magnetic moment of chromium(III) and cobalt(II) ions? - Chemistry Stack Exchange

The effective magnetic moments of Co2+ and Co3+ in SrTiO3 investigated by temperature-dependent magnetic susceptibility - ScienceDirect
Magnetic moment as a function of Cobalt measured at 4.2 K. The straight... | Download Scientific Diagram

SOLVED: Example 4: Consider the diamagnetic complex hexaamminecobalt(III)(+3). 1) What is the geometry of this ion complex? 2) Determine the hybridization of cobalt. 3) Calculate the spin-only magnetic moment of this complex.

Size effects in the magnetic anisotropy of embedded cobalt nanoparticles: from shape to surface | Scientific Reports

Q.28 (C) [Pd(CN),12- (D) [Nici,2- The spin magnetic moment of cobalt in the compound Hg(Co(SCN) 4) is - (A) V3 (B) 78 (C) Vī5 (D) 24
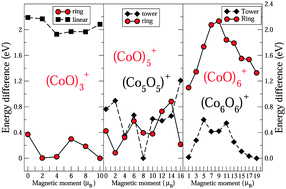
Structure, fragmentation patterns, and magnetic properties of small cobalt oxide clusters - Physical Chemistry Chemical Physics (RSC Publishing)






![The spin magnetic moment of cobalt in Hg[Co(SCN4)4] is : | Filo The spin magnetic moment of cobalt in Hg[Co(SCN4)4] is : | Filo](https://classroom-images.cdn.askfilo.com/classroom/1650264710045_rstgioom_1630410.jpg)


![The magnetic moment of cobalt of the compund Hg[Co(SCM)4] is [Given:Co+2.. The magnetic moment of cobalt of the compund Hg[Co(SCM)4] is [Given:Co+2..](https://storage.googleapis.com/filo-classroom-notes/thumb_classroom_29432591_XCIEK.jpeg)
![The spin magnetic moment of cobalt in the compound `Hg[Co(SCN)_(4)]` is - YouTube The spin magnetic moment of cobalt in the compound `Hg[Co(SCN)_(4)]` is - YouTube](https://i.ytimg.com/vi/Rrb9dYkq6OY/maxresdefault.jpg)

![The spin magnetic moment of cobalt in the compound Hg[Co(SCN),] is :- (1) 13 (2) V8 (3) V75 (1) 24 The spin magnetic moment of cobalt in the compound Hg[Co(SCN),] is :- (1) 13 (2) V8 (3) V75 (1) 24](https://toppr-doubts-media.s3.amazonaws.com/images/5605004/2a15c89c-4bff-4c66-a04d-1547f4f5eaec.jpg)