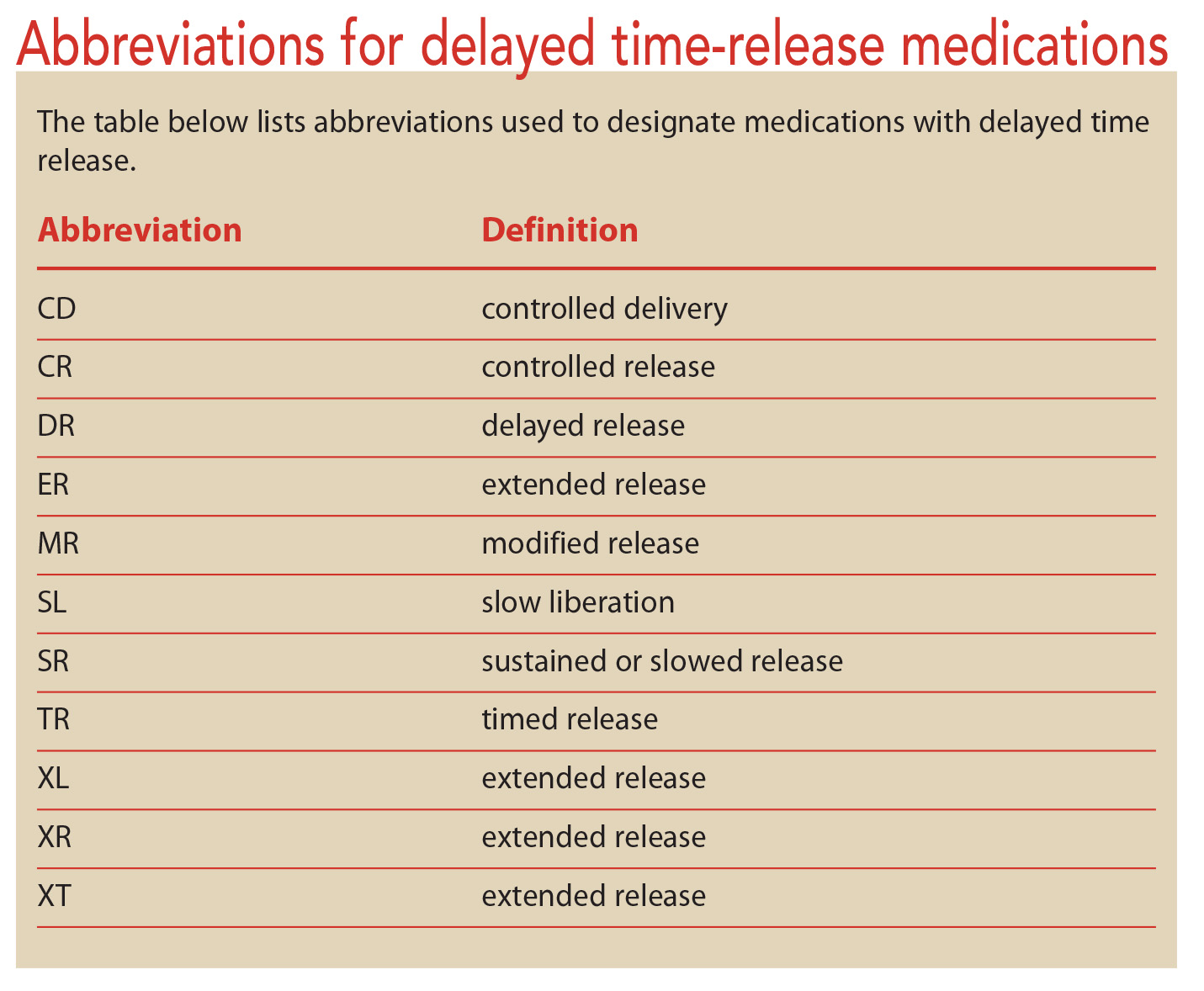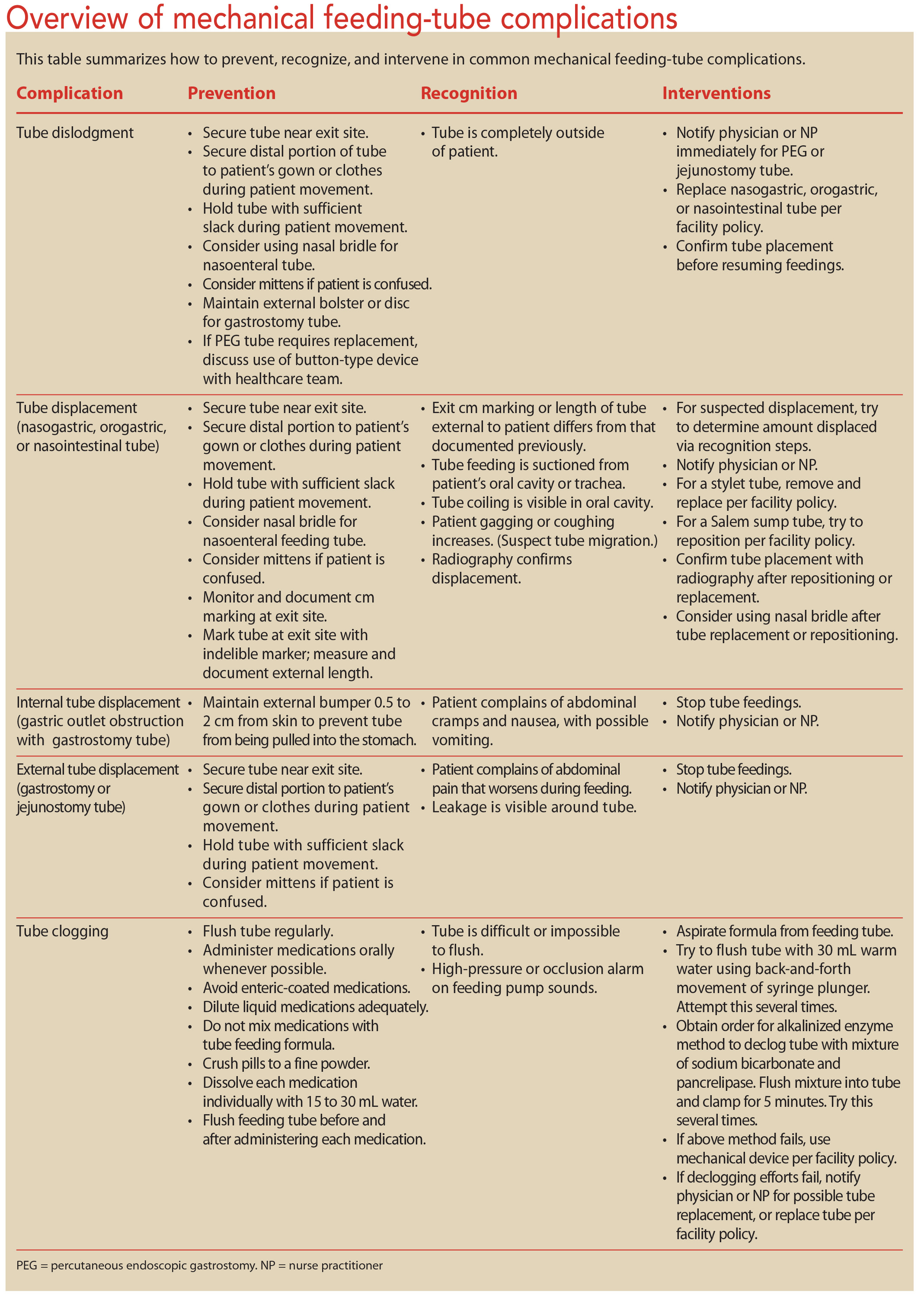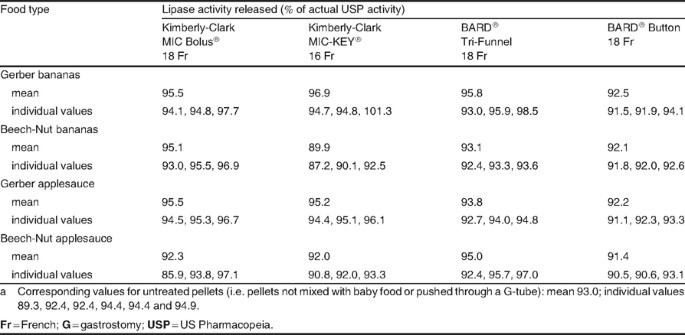
Administration of CREON® Pancrelipase Pellets via Gastrostomy Tube is Feasible with No Loss of Gastric Resistance or Lipase Activity | Clinical Drug Investigation
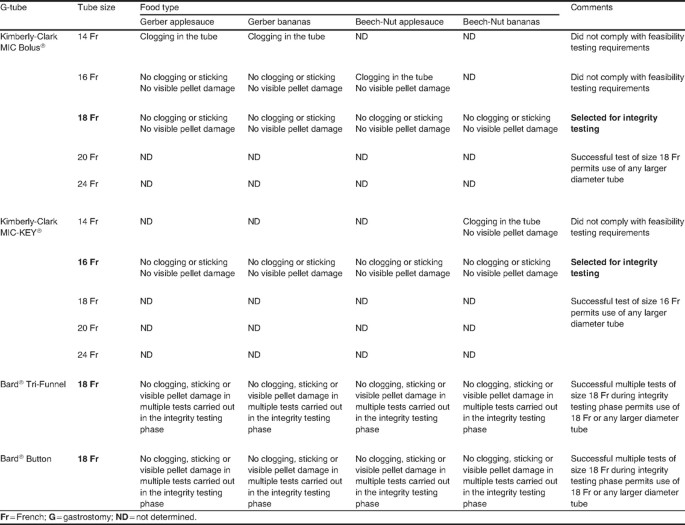
Administration of CREON® Pancrelipase Pellets via Gastrostomy Tube is Feasible with No Loss of Gastric Resistance or Lipase Activity | Clinical Drug Investigation