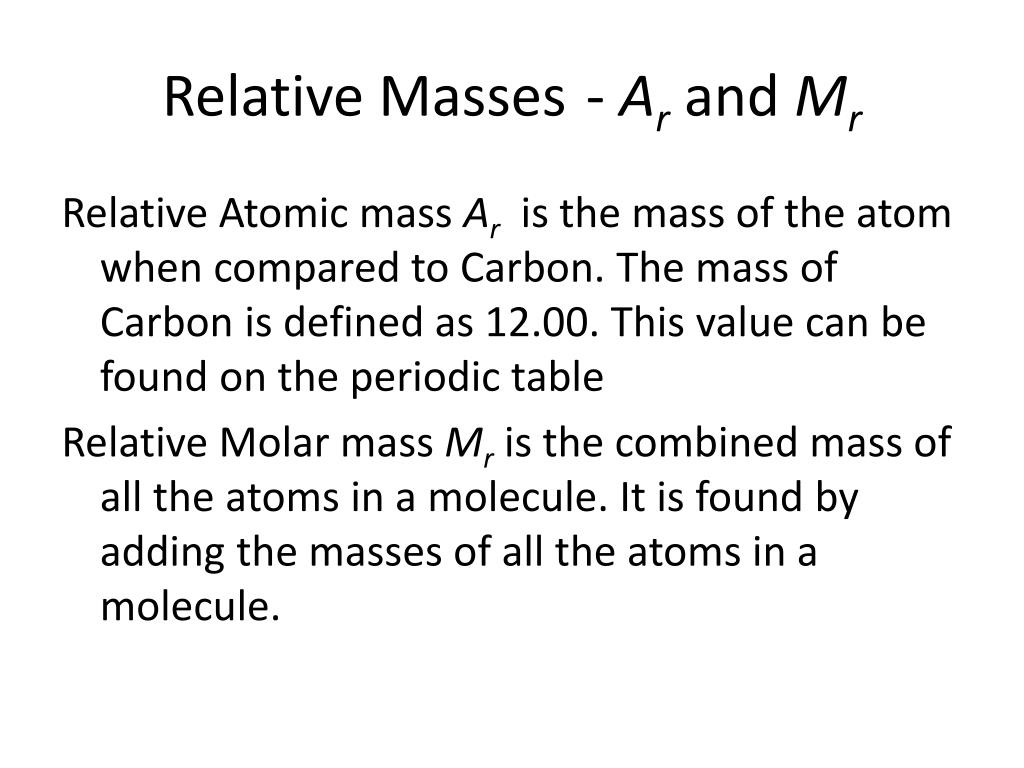Question Video: Calculating the Relative Molecular Mass of Xenon Difluoride from Its Chemical Formula | Nagwa
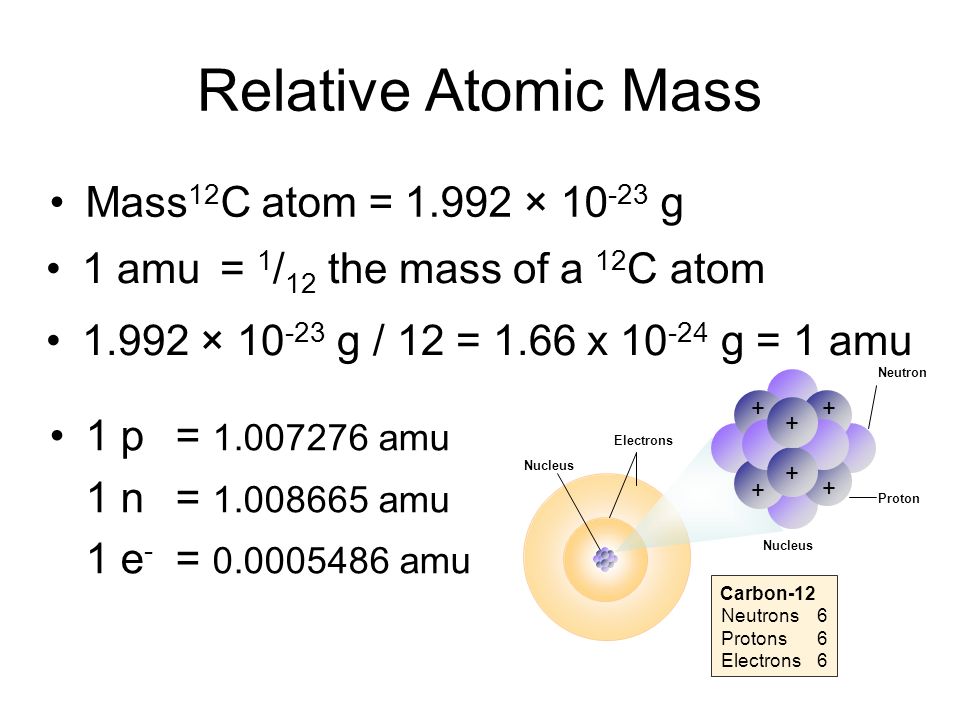
1.4 Isotopes, Radioisotopes, and Atomic Mass B3.1 explain the relationship between the atomic number and the mass number of an element, and the difference. - ppt download

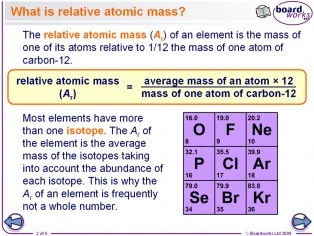
Defining how to calculate relative atomic mass of element relative isotopic mass definition gcse chemistry Calculations igcse O Level revision notes

Relative Atomic Mass Mass of atoms is incredibly small! So we have to use relative atomic mass. The symbol for relative atomic mass is Ar Carbon- 12 atom. - ppt download

Why only Carbon-12 is called as Relative Atomic Mass & Standard Atomic Mass |Why not other Elements💥 - YouTube
What's the math behind the similar numerical values of molar mass and relative molecular/atomic mass? - Quora
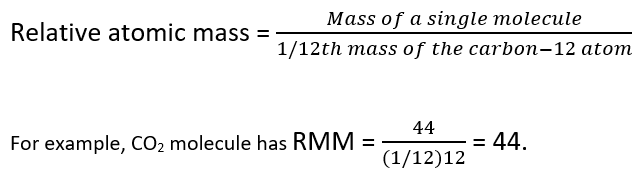
Relative Atomic Mass, Relative Molecular Mass & Mass Spectrometry | A-Level Chemistry Revision Notes

Relative atomic mass - A r Just another way of saying how heavy different atoms are compared with the mass of one atom of carbon – 12 (regular carbon!) - ppt download

The relative abundance of three isotopes of carbon C^(12), C^(13) and C^14 are 98.892%, 1.108% and 2 xx 10^(-10)% respectively. If the relative atomic masses of these isotopes are 12.00, 13.00335 and







:max_bytes(150000):strip_icc()/atomic-mass-and-mass-number-606105_v1-80df956ab98440bc9969531d1bb6c874.png)