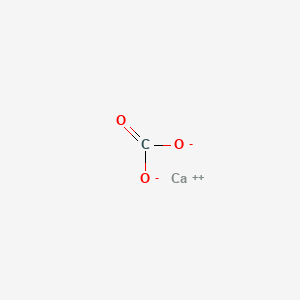
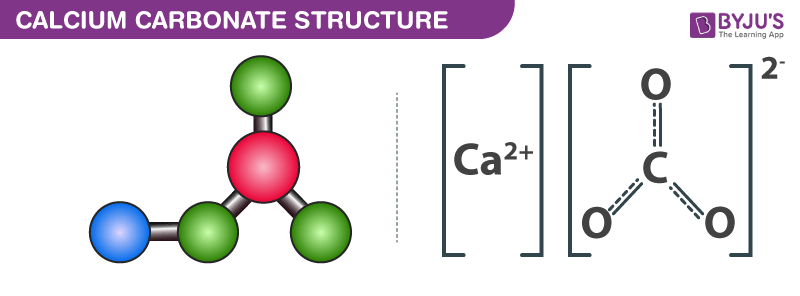
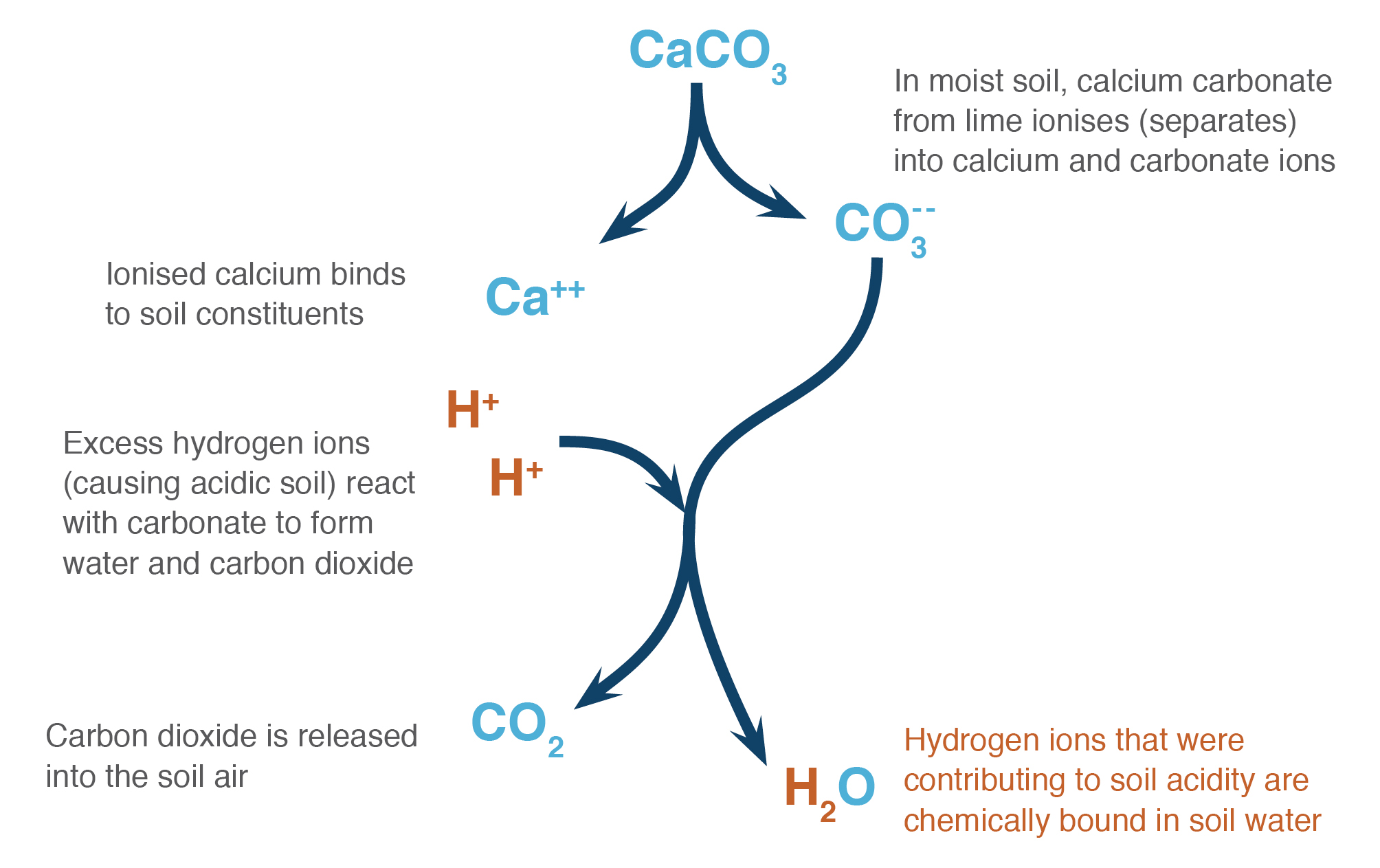
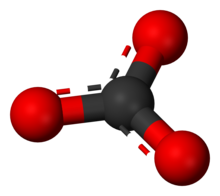
How would I go about creating a Lewis Dot structure for the Calcium Carbonate and Magnesium Hydroxide? I know how to do it for any compound with two elements such as SF5,
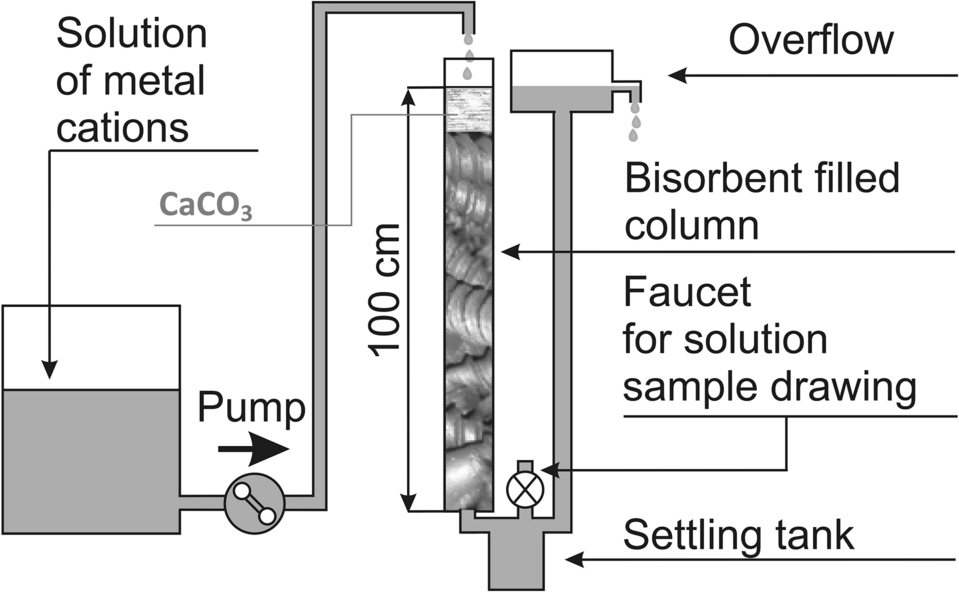
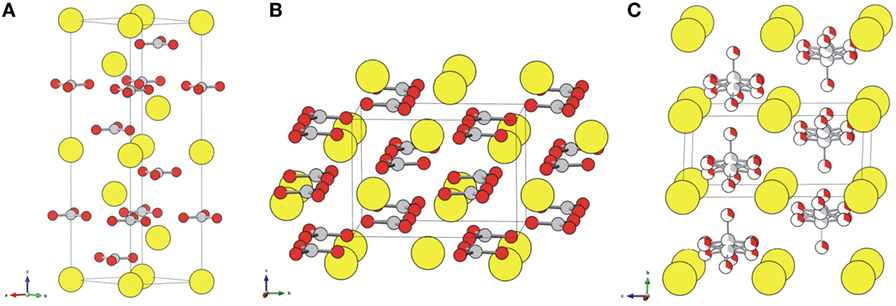
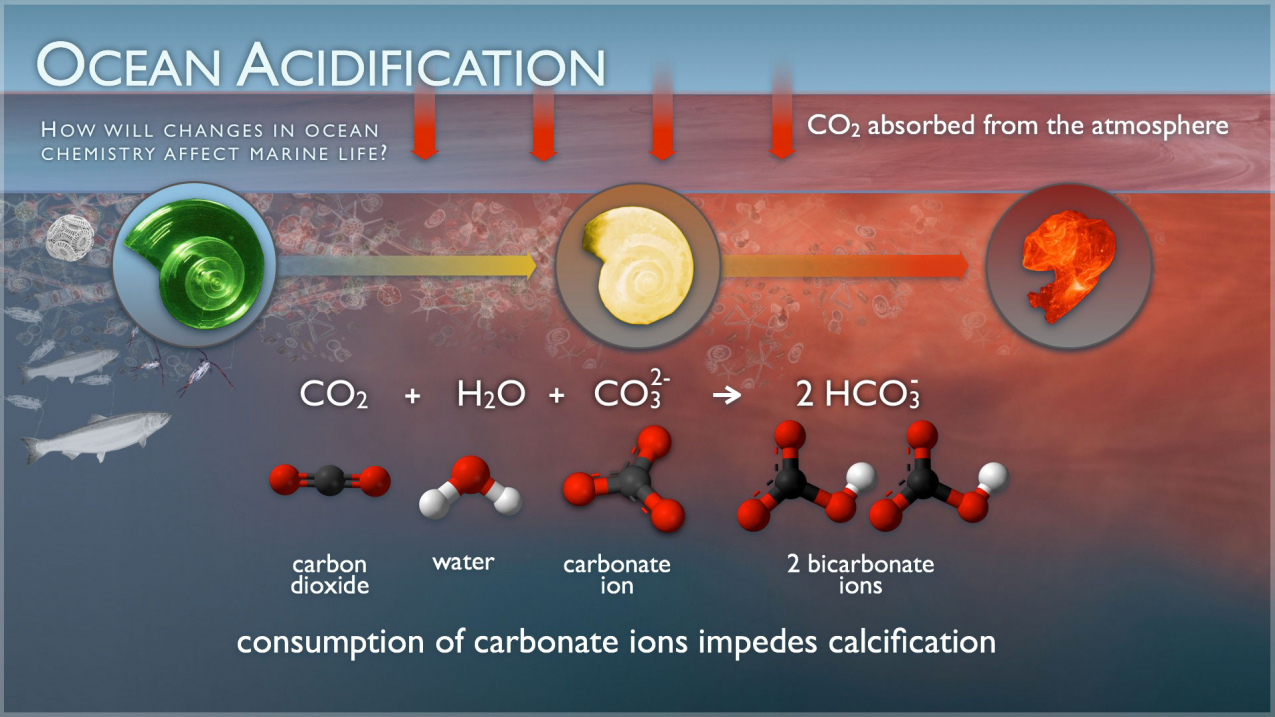
Role of calcium carbonate in the process of heavy metal biosorption from solutions: synergy of metal removal mechanisms | Scientific Reports
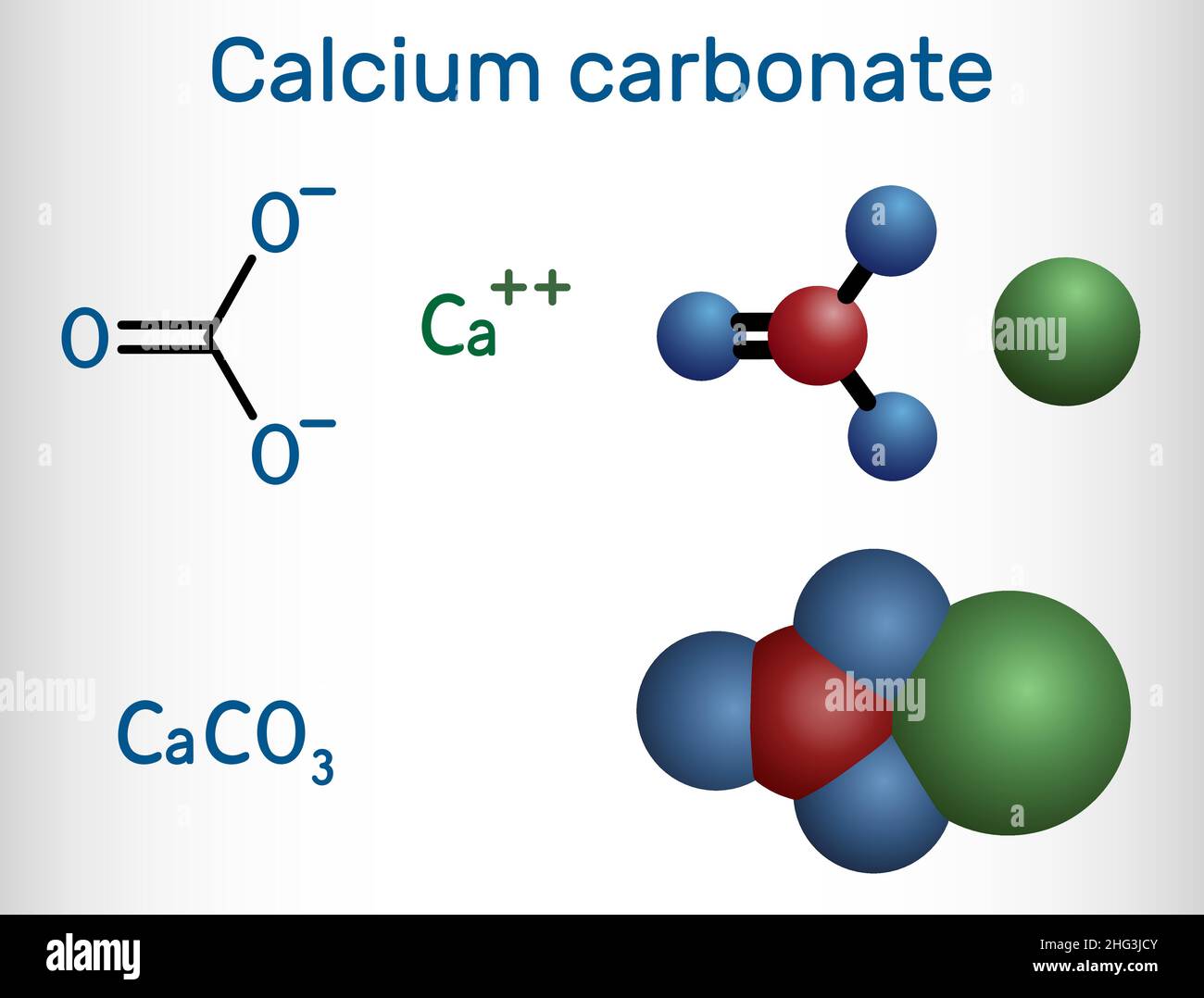
Calcium carbonate. Molecular model of the ionic mineral calcium carbonate (Ca.CO3). This chemical forms the main component of the shells of marine organisms and egg shells, and is an abundant mineral deposit
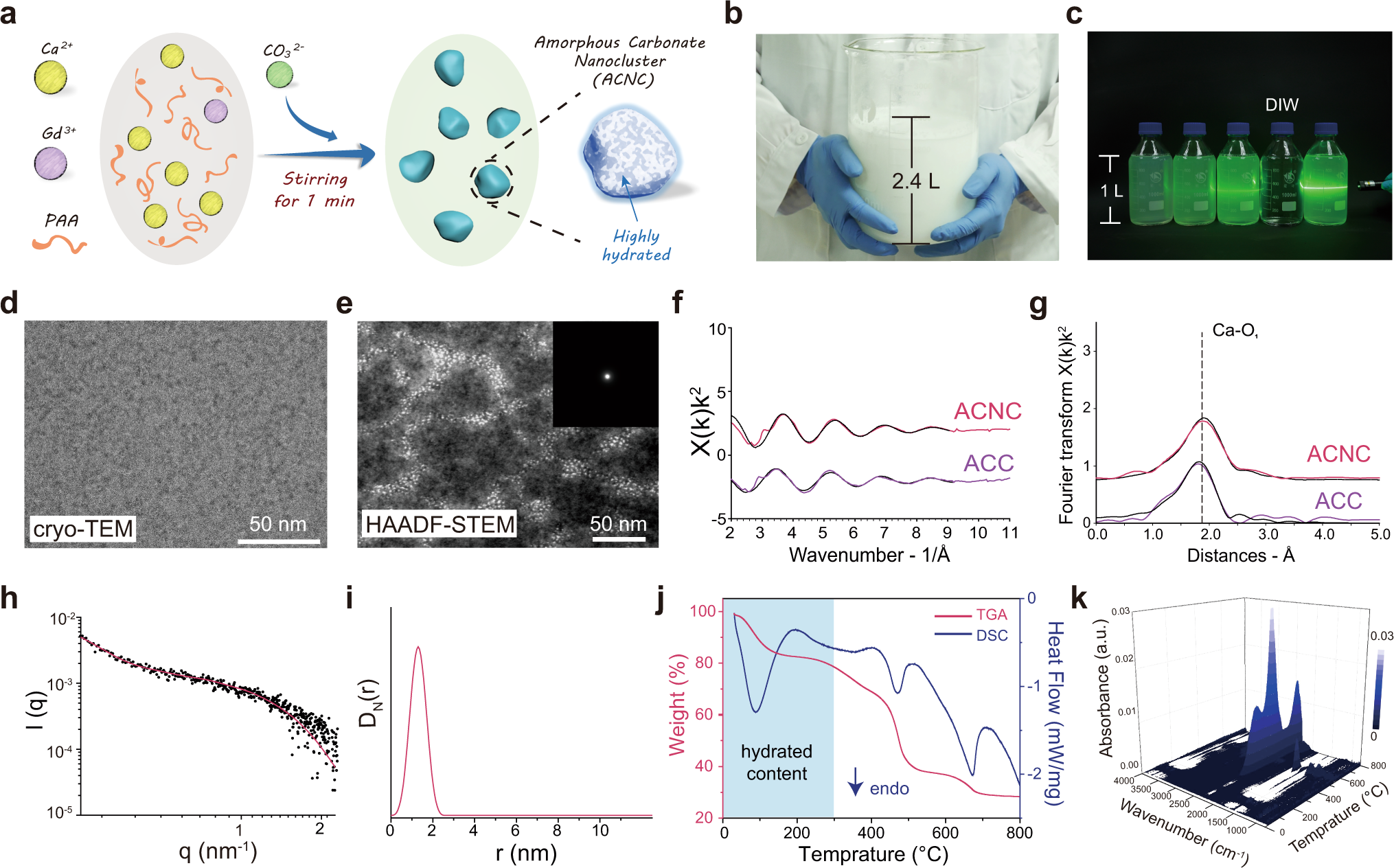
Highly hydrated paramagnetic amorphous calcium carbonate nanoclusters as an MRI contrast agent | Nature Communications